Overview
Our Catalysis platform seeks to discover, develop and apply new catalytic methods for the efficient and selective synthesis of biomedically relevant molecules.
The discoveries made in the Scheidt lab have allowed chemists to access new modes of reactivity that have been inaccessible through traditional approaches. We have pioneered the use of N-heterocyclic carbenes (NHCs) in catalysis to achieve Umpolung reactivity, allowing us to access a host of catalytically generated nucleophiles. We have also developed a set of novel planar chiral NHC ligands by incorporating a fused ferrocene motif, providing a unique steric environment. Currently, our lab is exploring the combination of NHCs with single electron processes driven by light. Our continuing goal is to integrate different catalysis modes in cooperative strategies to expand the NHC field and ultimately provide the community with powerful complexity generating tools.
Personal Development
To advance your scientific knowledge and understanding at the highest level.
Reaction Discovery
Become an expert in catalytic method development using state of the art research technologies including high throughput reaction screening.
Interdisciplinary
Embrace creative thinking to solve challenging synthetic problems in multiple fields of catalysis including photocatalysis, biocatalysis, and transition metal catalysis.
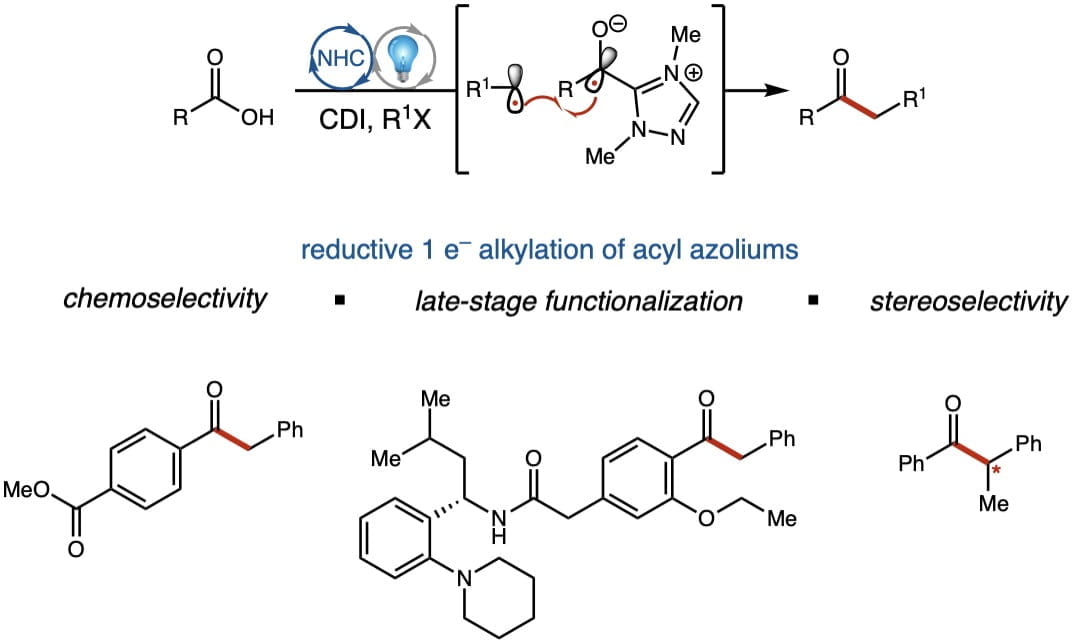
NHC Photoredox Platform
Recently, our lab has been interested in merging single electron chemistry with NHC catalysis. Using high throughput reaction screening, we were able to develop a novel photocatalytic reduction of an acyl azolium to generate an azolium radical species. This azolium radical can then undergo radical-radical coupling with various radical sources providing substituted ketone products, and could be applied to the late-stage functionalization of pharmaceuticals. By harnessing the power of light, we are continuing to push the boundaries of current NHC chemistry to discover new and exciting reactions.
Previous Endeavors
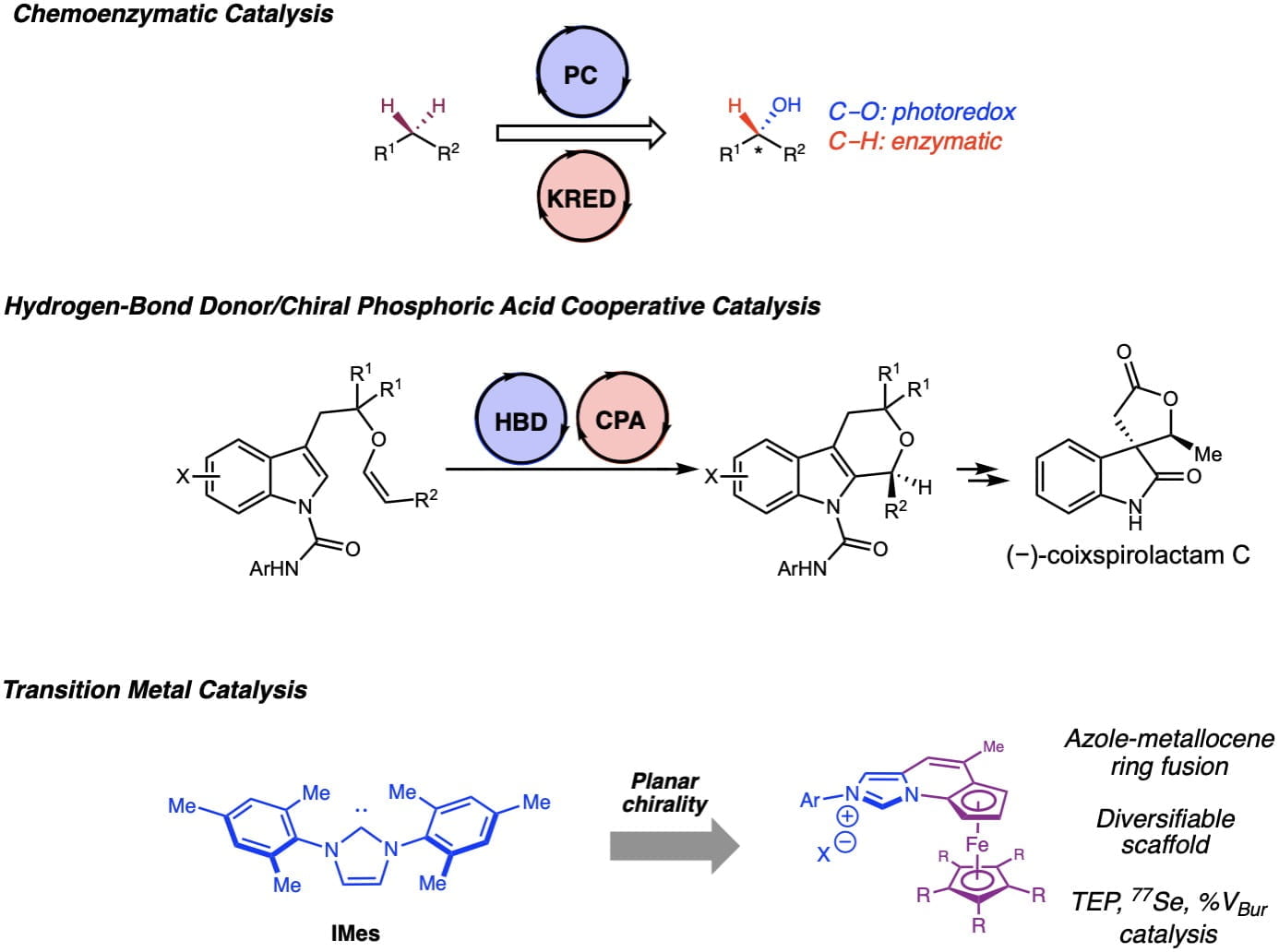
Our method development efforts seek to address a wide range of synthetic challenges. These efforts have allowed us to leverage knowledge acquired in reaction development towards the synthesis of small molecules relevant to cancer biology and psychopharmacology to drive the advancement of new therapeutics. Recently, our lab developed a new class of novel planar chiral NHC ligands that could be used to synthesize medicinally releveant enantioenriched a-amidoboronates. We have also developed new methods merging hydrogen-bonding catalysis with chiral phosphoric acids for enantioselective oxa-Pictet-Spengler reactions, as well as merging photocatalysis with biocatalysis for C-H functionalization.
Latest Publications
The Scheidt lab has a strong emphasis on creative problem-solving and a commitment to mentorship.
View All Publications ›

Photochemical phosphorus-enabled scaffold remodeling of carboxylic acids
Peng, Q.; Hwang, M. U.; Rentería-Gómez, A.; Mukherjee, P.; Young, R. M; Qiu, Y.; Wasielewski, M. R.; Gutierrez, O.; Scheidt, K. A.* Science 2024, 385, 1471-1477.
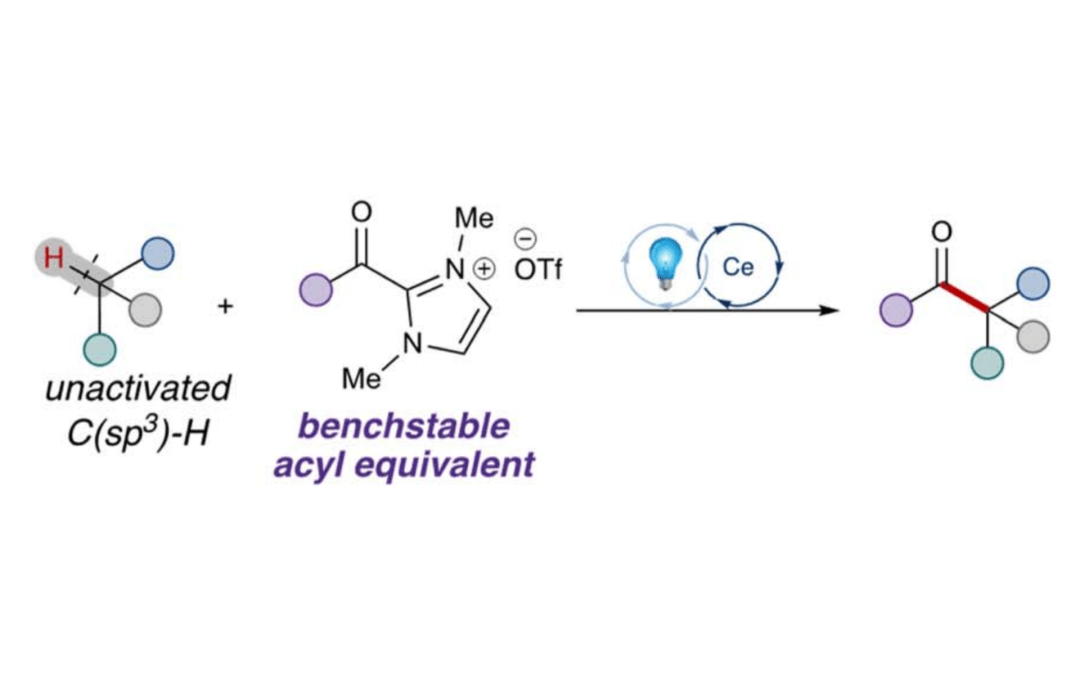
Photoinduced cerium-catalyzed C–H acylation of unactivated alkanes
Cao, J,; Zhu, J. L.; Scheidt, K. A.* Chem. Sci. 2023, 15, 154-159.
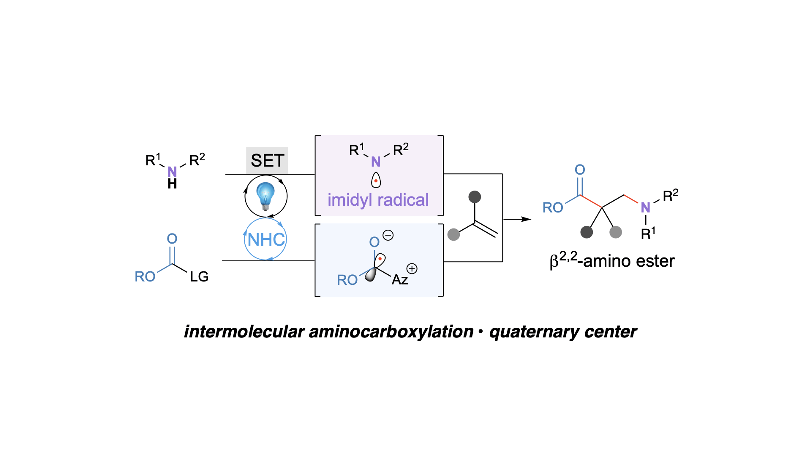
Cooperative Carbene Photocatalysis for β-Amino Ester Synthesis
Tanaka, N.; Zhu, J. L.; Valencia, O. L.; Schull, C. R.; Scheidt, K. A.* J. Am. Chem. Soc. 2023, 145, 24486-24492.